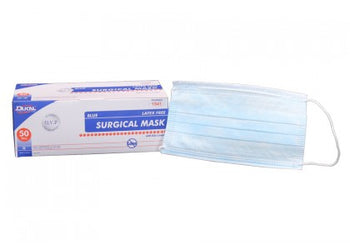
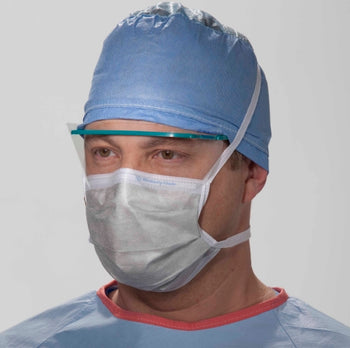
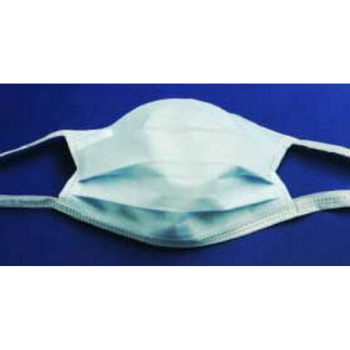
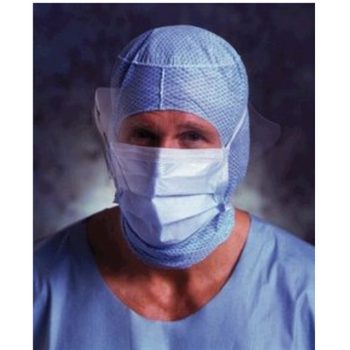
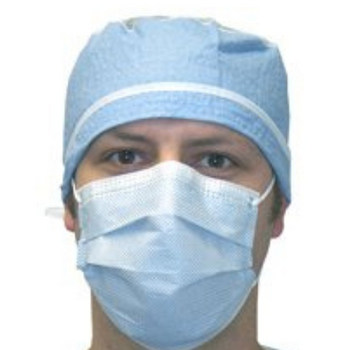
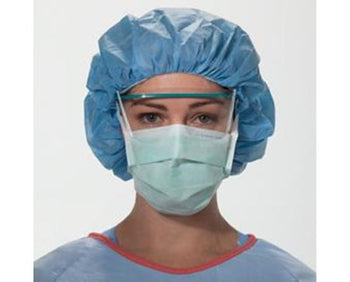
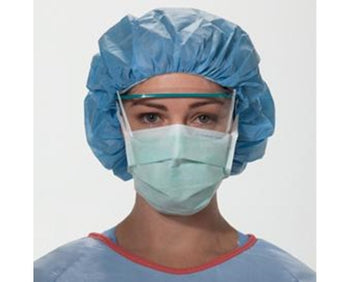
Columbia 600 Electrode...
The Columbia 600 Electrode is for use with all NMES devices that treat Dysphagia patients. It is FDA cleared for use on the neck, and to be used with primary Dysphagia treatment devices. All electrodes are single patient-multiple use application, and low impedance. The carbon mesh constructio...
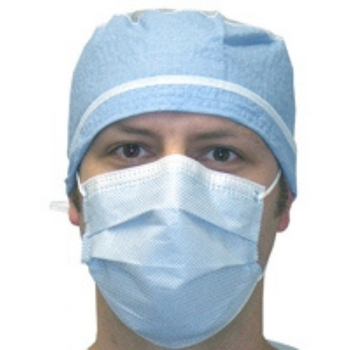
Columbia 600 Electrode...
The Columbia 600 Electrode is for use with all NMES devices that treat Dysphagia patients. It is FDA cleared for use on the neck, and to be used with primary Dysphagia treatment devices. All electrodes are single patient-multiple use application, and low impedance. The carbon mesh constructio...
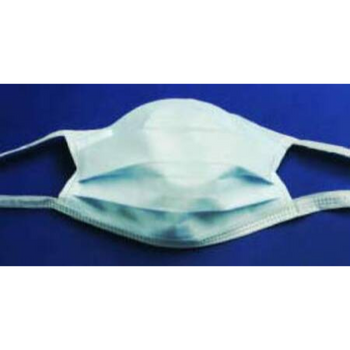
Columbia 600 Electrode...
The Columbia 600 Electrode is for use with all NMES devices that treat Dysphagia patients. It is FDA cleared for use on the neck, and to be used with primary Dysphagia treatment devices. All electrodes are single patient-multiple use application, and low impedance. The carbon mesh constructio...
Columbia 600 Snap Electrode...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600 Snap Electrode...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600 Snap Electrode...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600 Snap Electrode...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600B Electrodes...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600B Electrodes...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600B Electrodes...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...
Columbia 600B Electrodes...
Used with all NMES devices that treat Dysphagia patients, the FDA approved the elcetrodes for use on the neck and to be used with primary Dysphagia treatment devices. All the electrodes are single patient-multiple use application. For added comfort and strength, the electrodes are made from a...